—Barrington Williams, B1Daily
In 1951, a 31-year-old Black woman named Henrietta Lacks walked into Johns Hopkins Hospital seeking treatment for aggressive cervical cancer. What happened next would transform modern medicine — and ignite decades of ethical debate over consent, race, and profit in biotechnology.
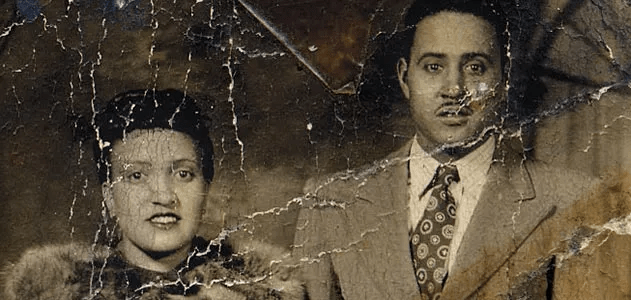
Without her knowledge or permission, doctors removed samples of her cancer cells during treatment. Unlike other human cells scientists had tried to grow in laboratories, Lacks’ cells did something extraordinary: they replicated indefinitely. They became the first immortal human cell line, later named HeLa, derived from the first two letters of her first and last name.
The Birth of a Biomedical Revolution
HeLa cells quickly became one of the most important tools in medical research. They were used to develop the polio vaccine, advance cancer treatments, study viruses such as HIV, test the effects of radiation, and even travel into space to examine how human cells react to zero gravity. To this day, HeLa cells remain foundational in laboratories around the world.
Yet while pharmaceutical and biotechnology companies built entire industries around these cells — producing, distributing, and profiting from them — Henrietta Lacks’ family lived in poverty for years, unaware that part of their mother’s biological material had become one of the most valuable tools in science.
Consent and Exploitation
At the time her cells were taken, U.S. law did not require doctors to obtain informed consent for the use of discarded tissue in research. The practice was common, but Lacks’ case became emblematic of broader patterns of medical exploitation, particularly involving Black patients in the mid-20th century.
The ethical controversy intensified as HeLa cells became commercialized. Biotechnology companies mass-produced and sold the cells globally, generating significant revenue. Meanwhile, Lacks’ descendants often struggled to access adequate healthcare themselves.
Critics argue that her case reflects systemic inequities in medical research — where marginalized patients historically bore the risks while institutions reaped the rewards.
Legal and Ethical Aftermath
Over time, the story of Henrietta Lacks prompted major conversations about bioethics. Informed consent standards were strengthened. Institutional Review Boards (IRBs) became standard in overseeing research involving human subjects. Discussions around ownership of genetic material intensified.
In 2013, after scientists published the HeLa genome without consulting the family, negotiations led to an agreement granting Lacks’ descendants limited control over access to her genetic data. More recently, in 2023, a biotech company reached a confidential settlement with the Lacks family in a lawsuit alleging unjust enrichment from the commercialization of HeLa cells.
Legacy Beyond the Lab
Henrietta Lacks’ story gained global recognition through The Immortal Life of Henrietta Lacks by Rebecca Skloot and its later film adaptation. Her legacy now stands at the crossroads of scientific triumph and ethical reckoning.
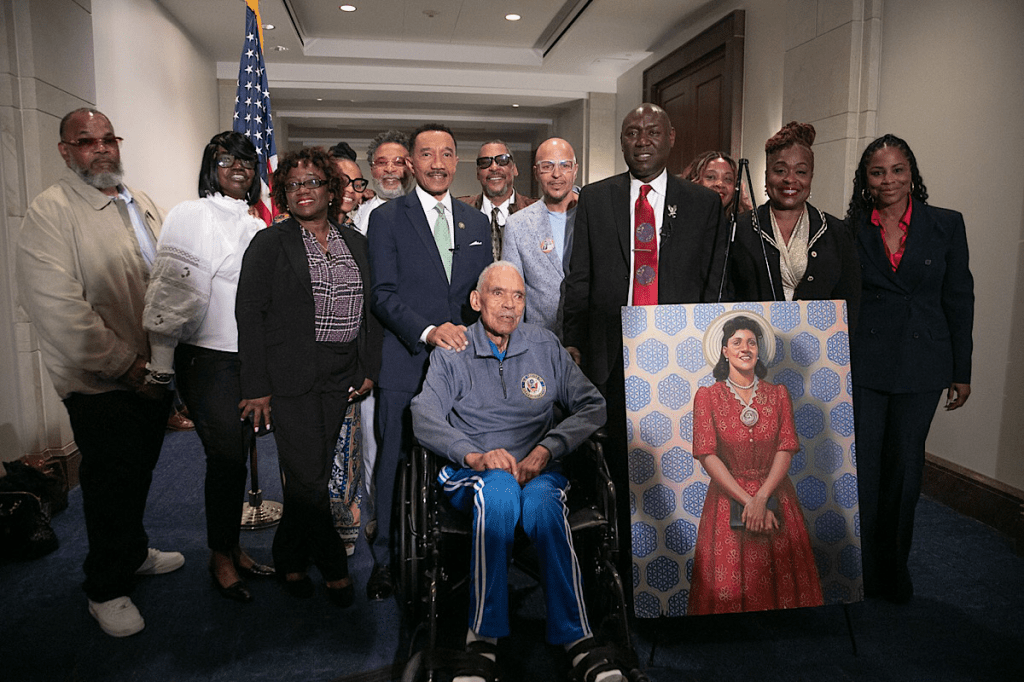
Today, HeLa cells symbolize both extraordinary medical progress and the cost at which some breakthroughs were achieved. Lacks never consented to her cells being taken, yet they helped save millions of lives.
Her story forces a difficult but necessary question: Who owns the building blocks of the human body — the individual, the institution, or the marketplace?
More than seventy years later, Henrietta Lacks remains central not just to biology textbooks, but to ongoing debates about race, equity, and the ethics of profit in modern biotechnology.
—Barrington Williams, B1Daily





Leave a comment